Escherichia coli Pre and Post-weaning Diarrhoea
|
Causal
agent |
Escherichia
coli – E. coli. A gram negative rod bacteria |
|||
|
Age group |
From
birth to around 10 days of age. First
few weeks post-weaning
|
|||
Clinical signs
|
||||
|
0-3 days |
Sudden death, with very few clinical signs. Possibly some diarrhoea |
|||
|
3-10 days |
Acute and chronic diarrhoea, pasty yellow colour. Piglets may be found dead, but most have
clinical signs which lead to dehydration and ultimately death. |
|||
Post-weaning
|
Acute and chronic diarrhoea. Weaners may demonstrate ill-thrift. The diarrhoea progressively leads to
dehydration and death. |
|||
|
|
|
|
||
|
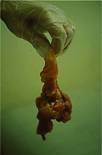
Neonatal death |
Diarrhoea in farrowing |
Piglet with E. coli diarrhoea |
||
|
|
|
|
||
|
Diarrhoea on the walls |
Diarrhoea post-weaning |
E. coli diarrhoea in weaners |
||
Transmission
|
||||
|
E. coli are ubiquious (everywhere). Some types are more pathogenic depending on
fimbiae. The organism may be easily
transmitted around the farm. |
||||
|
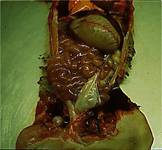
Post-mortem Lesions |
||||
|
There may be very few gross pathological findings. Dilated swollen small intestines fluid
filled. Sometimes congestion of the
stomach or small intestines may be seen.
The intestinal contents will be alkaline. |
||||
|
|
|
|||
|
Gross-postmortem
findings dilated small intestines and stomach |
Loops
of bowel with very watery alkaline diarrhoea |
|||
|
Diagnosis |
|||
|
|
Drawing of the classification of Escherichia coli Pathogenic swine E.
coli are normally classified by: Have one of the following four fimbriae (attachments) F4
(K88), F5 (K99), F6 (987P) or F41 Haemolytic. Produce
toxins (exotoxins) – LT, Sta or STb. Most E. coli
problems are associated with management and environmental factors, in
particular draughts. Examination of
the environment is an essential component to any diagnosis. |
||
|
As soon as scour starts, submit rectal swabs |
|||
|
If piglets die, submit piglet for post-mortem examination |
|||
|
If scour continues, submit live piglets before treatment |
|||
|
Treatment |
|||
Individual litters
|
Treat
whole litter as soon as one piglet starts to show symptoms |
||
|
Place
a trough drinker filled with water, electrolytes and glucose. This must be replaced at least 4 times
daily. Keep clean |
|||
|
If
creep is provided, remove the creep |
|||
|
All
piglets to be given colostrum |
|||
|
Administer
antimicrobial oral dose measured doses once a day
by mouth, see the practice. Ensure you
do not use this medicator for healthy piglets |
|||
|
Inject
with a suitable antimicrobial. |
|||
|
Clean
up diarrhoea as soon as possible |
|||
|
The
stockpeople must wash their hands after treating the piglets and dip their
boots in disinfectant |
|||
|
Syringes
or oral dosers must not be used on a sick piglet and then used on a healthy
piglet |
|||
|
If
diarrhoea occurs after day 10 consider coccidiosis |
|||
|
Cross-fostering |
Piglets
movements must be carefully monitored.
A lot of scouring is associated with fostering. Review cross-fostering protocols |
||
|
Utensil movement |
All
rooms should have their own brush and shovel |
||
|
Each
brush and shovel should be placed in disinfectant when not in use. Ensure disinfectant is still working |
|||
|
Teeth
clipping, tattooing, notching and tail docking equipment must be thoroughly cleaned
between rooms |
|||
|
Ensure
that the rooms are thoroughly cleaned between batches |
|||
|
Consider
lime-washing to enhance hygiene and disinfection |
|||
Control
|
Pressure
washing principles must be exemplary |
||
|
Practice
lime washing if possible |
|||
|
Practice
all-in/all-out – pig flow |
|||
|
If
diarrhoea is a persistent problem have the environment examined in detail
i.e. for draughts |
|||
|
Ensure
preventative protocols are adhered to, i.e. vaccination and feed-back |
|||
|
F4
genetically resistant pigs are available but not used commercially. Do not select from sows/gilts who have any history of diarrhoea
in the farrowing house. Over time this
will select away from susceptibility towards E. coli types especially F4. |
|||
|
Common
differentials |
|||
|
Viral diarrhoea – normally acid intestinal contents. Coccidiosis |
|||
|
Zoonosis |
|||
|
E. coli has the potential of being zoonotic |
|||
Pre-weaning
Scour Protocols
|
Piglet
treatments |
|
|
1 |
Treat whole litter as soon as one piglet starts
to show symptoms |
|
2 |
Place a cube drinker filled with water,
electrolytes and glucose. This must be
replaced at least 4 times daily. Keep
clean |
|
3 |
If creep is provided, remove the creep |
|
4 |
All piglets to be given colostrum |
|
5 |
Administer antimicrobial
oral dose measured
doses once a day by mouth, see the practice.
Ensure you do not use this medicator for healthy piglets |
|
6 |
Inject with a
suitable antimicrobial. |
|
7 |
Clean up diarrhoea as soon as possible |
|
8 |
The stockpeople must wash their hands after
treating the piglets and dip their boots in disinfectant |
|
9 |
Syringes or oral dosers must not be used on a sick
piglet and then used on a healthy piglet |
|
10 |
If diarrhoea occurs after day 10 consider
coccidiosis |
|
Cross-fostering |
|
|
1 |
Piglets movements must be carefully
monitored. A lot of diarrhoea is
associated with fostering. Review
cross-fostering protocols |
|
Movements
of utensils and people from one room to the next |
|
|
2 |
All rooms should have their own brush and shovel |
|
3 |
Each brush and shovel should be placed in
disinfectant when not in use. Ensure
disinfectant is still working |
|
4 |
Teeth clipping, tattooing, notching and tail
docking equipment must be thoroughly cleaned between rooms |
|
5 |
Ensure that the rooms are thoroughly cleaned
between batches |
|
6 |
Consider lime-washing to enhance hygiene and
disinfection |
|
Investigation |
|
|
1 |
As soon as diarrhoea starts, submit rectal swabs |
|
2 |
If piglets die, submit piglet for post-mortem
examination |
|
3 |
If diarrhoea continues, submit live piglets before
treatment |
|
Long
term |
|
|
1 |
Pressure washing principles must be exemplary |
|
2 |
Practice lime washing if possible |
|
3 |
Practice all-in/all-out – pig flow |
|
4 |
If scour is a persistent problem have the
environment examined in detail i.e. for drafts |
|
5 |
Ensure preventative protocols are adhered to,
i.e. vaccination and feed-back |
Classic “causes” of pre-weaning
diarrhoea
|
1 |
A range of disease agents – E. coli, TGE, Coccidiosis, Clostridia,
Rotovirus etc. |
|
2 |
Almost any air movement is undesirable >0.2 m/sec (> 50
feet/min) is a draught |
|
3 |
Chilling of the piglets, check lying patterns and creep
temperatures (ideally 30•C) |
|
4 |
Variable temperatures in
the creep |
|
5 |
Damp floors particularly in
the creep area |
|
6 |
Poor colostrum intake |
|
7 |
No milk in the sows, check
udder line – mycotoxins and management |
|
8 |
Degree of cross-fostering |
|
9 |
Piglet treatments not been clean enough,
Check cross-contamination between healthy and sick piglets |
|
10 |
Infection transfer - is
there a separate brush and scrape for each room, foot baths, personal hygiene |
|
11 |
Poor room cleaning between
batches |
|
12 |
Number of sows farrowing each week, application of all-in
all out and pig flow |
|
13 |
Presence of udder oedema |
|
14 |
Amount of navel bleeding |
|
15 |
Type of iron injection utilised, more post-weaning scour |
|
16 |
Vaccine storage protocols |
As part of the investigation, if post-mortem examinations are required, select an acutely sick piglet, not a chronic piglet which is likely to have secondary infections which may mask the actual causal agent(s).